AI-powered early detection for dialysis vascular access failure
ShuntWizard analyzes simple sound recordings from AV fistulas (via smartphone or digital stethoscope) to flag early signs of stenosis—before thrombosis or emergency hospitalization occur.
The Problem
The fragile lifeline of hemodialysis
Vascular access dysfunction is common, often detected late, and leads to costly interventions, catheter use, and hospitalizations. Current monitoring relies on subjective auscultation and resource-intensive ultrasound—leaving a gap for standardized, scalable early detection.
Complications
Up to 50% of fistulas develop stenosis
Arteriovenous fistula dysfunction is the leading cause of hospitalization in dialysis patients, yet early signs are routinely missed.
Hospitalizations
Most complications detected too late
Current monitoring relies on subjective auscultation and resource-intensive ultrasound, leaving a gap for standardized, scalable early detection.
Consequences
Late detection triggers urgent interventions
Thrombosis leads to emergency access events, temporary catheters, and increased infection risk—all of which are largely preventable.
Cost Impact
$1B+ annual burden in the US alone
Vascular access complications are a major driver of the total cost of dialysis care, with enormous system-wide pressure to reduce preventable events.
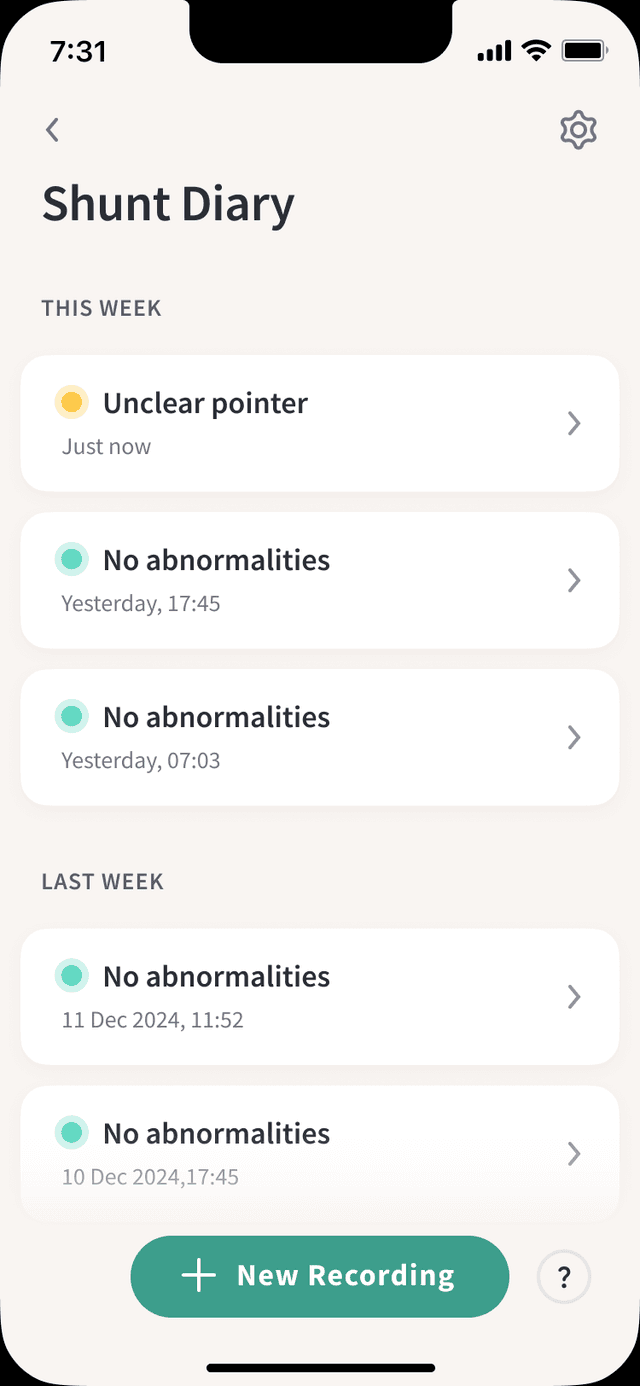
How It Works
Turn a 10-second sound recording into an actionable risk insight
From recording to diagnosis in under 30 seconds.
Record
Hold your smartphone near your shunt for 10 seconds. Or use a digital stethoscope at the dialysis center. No special training required.
Analyze
AI processes the acoustic signal using validated signal processing and AI to detect abnormal patterns in the shunt sound.
Act
Get an instant risk result. Abnormal patterns are flagged for earlier clinical follow-up—giving time to act before thrombosis occurs.
Our Products
Built for both patients and health care professionals
Patient App
Home Monitoring
- Self-checks in under a minute
- Trend-based risk detection over time
- Designed for remote care workflows
- Alerts forwarded to treating shunt center
.png&w=640&q=75)
Professional Tool
Dialysis Center
- Standardized screening during routine care
- Rapid point-of-care decision support
- Integrates into center workflows
- Analysis within seconds
.png&w=640&q=75)
Traction
Clinical data incoming!
We are building with clinical rigor from the start—real data, real patients, real outcomes. Interested in pilots or investment?
Clinically Labeled Data Collection Underway
Active data collection with partner dialysis centers. Building the proprietary dataset needed for model training and clinical validation.
Regulatory-Grade Development Process
Building toward a regulated medical product from day one. Documentation, risk management, and quality processes designed for regulatory approval.
Core Patent Filed
Patent filed covering the core method and workflow for sound-based shunt monitoring. Continuing patent strategy in development.
Prototype Apps and Pipelines in Development
Patient app and professional tool prototypes actively being built. Signal processing and AI pipelines under validation.
Market Timing
A rare window: clinical need + AI readiness
Hardware Independent
Commodity microphones and digital stethoscopes are now everywhere. The infrastructure already exists—we just need the intelligence layer.
AI Maturity
AI makes sound-based, signal-based detection scalable and consistent. Deep learning now enables reliable acoustic pattern recognition at scale.
Shifting Kidney Care
Kidney care is shifting toward prevention and value-based models. Payers and providers are actively seeking scalable early-detection solutions.
Major Cost Driver
Dialysis access complications remain one of the largest avoidable cost drivers in chronic care—creating strong financial incentive for innovation.
Competitive Advantage
Built to compound
Our moat deepens over time. Every patient adds data, every integration deepens workflow lock-in, and every regulatory milestone raises the barrier to entry.

Proprietary Longitudinal Dataset
Clinically labeled acoustic data collected with ground truth from partner centers. Trained on real patient data—not public datasets.
Workflow and Product Moat
Two-sided product strategy (patient + provider) creates compounding lock-in. Deep integration into existing care workflows.
Regulatory-Grade Engineering
Built in preparation for MDR/FDA submission from day one. Risk management, documentation, and quality systems create high barriers for fast-followers.
Patent Protection
European Patent Application No. 25182295.3 filed, covering the fundamental method and workflow. Continuing patent strategy under development to protect key innovations.
Vision
Building the future of vascular access monitoring
We're starting with dialysis vascular access—a critical and underserved clinical need. Our vision is to build the leading sound-based monitoring platform for chronic vascular care.
Team and Advisors
Built by people who ship in regulated healthcare

Aleksandra Milisavljevic
CEO & Co-Founder
Aleksandra is a tech entrepreneur and product leader focused on building practical, real-world health-tech solutions. As Co-Founder of Shunt Wizard, she helps translate complex clinical needs into clear, usable digital products that support better care and decision-making. She also leads Inviggo, a global software outsourcing company serving healthcare and fintech clients, and founded Selfnest, a mental health awareness product
LinkedIn
Stasys Hiob
CTO & Co-Founder
Stas is an experienced technology leader and CTO with a strong track record of building and scaling regulated health-tech products. He excels at turning complex clinical needs into real-world, certified digital solutions by leading teams with clarity and purpose. Passionate about impactful innovation, Stasys drives technology strategy and execution to deliver reliable, compliant healthcare platforms that improve outcomes.
LinkedInStatements from Key Opinion Leaders
Early detection of stenosis is the single most impactful intervention we can make for dialysis patient outcomes. ShuntWizard’s approach is clinically sound and fills a genuine gap.

Dr. med. Meyer
Chairman Committee for Vascular Access and Interventional Nephrology (German Society of Nephrology)
The combination of acoustic monitoring and AI has significant potential to transform how we approach vascular access surveillance—moving from reactive to proactive care.

Prof. Hyhlik-Dürr
Vascular Surgery, Medical Vaculty, University of Augsburg
FAQ
Questions
Soon — ShuntWizard is being built as a regulated medical device from day one. We follow a rigorous regulatory development process including risk management, clinical evidence generation, and quality management. We are not yet CE Marked or FDA cleared, and the current prototypes are for research/validation use only.
Validation is built on clinically labeled acoustic data collected with partner dialysis centers — real patient recordings matched to clinical ground truth (imaging, intervention outcomes). We are conducting prospective validation studies to establish sensitivity, specificity, and clinical utility before seeking regulatory approval.
The primary target payers are dialysis providers and kidney care programs who benefit from reducing emergency interventions and hospitalizations. We are initially focusing on pilot partnerships with dialysis centers to demonstrate clinical value and ROI. Insurance reimbursement pathways will be pursued alongside CE Mark / FDA clearance.
Have more questions? .
Let's talk
Whether you're interested in a pilot partnership, investment, or clinical collaboration — we'd love to hear from you.
Join dialysis centers and nephrology teams improving patient outcomes